Steeping is the final step in the lives of tea leaves. And in their final act, they slowly unfold and unravel, creating a beverage that tells the story of where they came from. Every time we drink liquor from the steeped leaves, it tells us what the weather was like before they were plucked and how they were handled, processed, and stored before they reached your cup.
Chemically speaking, steeping refers to the act of infusing tea leaves in a solvent (water) to make a solution that is on average 98% water and 2% compounds from within the tea leaves.
How Steeping Works
But what actually happens during steeping? When tea leaves are added to water, they absorb some of it and become rehydrated. This absorption of water into the tea leaves allows for the initiation of steeping, the process of extracting the soluble compounds from the tea leaves and dissolving them in water. The driving force of the steeping process is the difference in concentrations of dissolved compounds in the leaves and the water. Compounds in the leaves move from an area of high concentration to an area of low concentration until equilibrium is achieved, a process called diffusion.
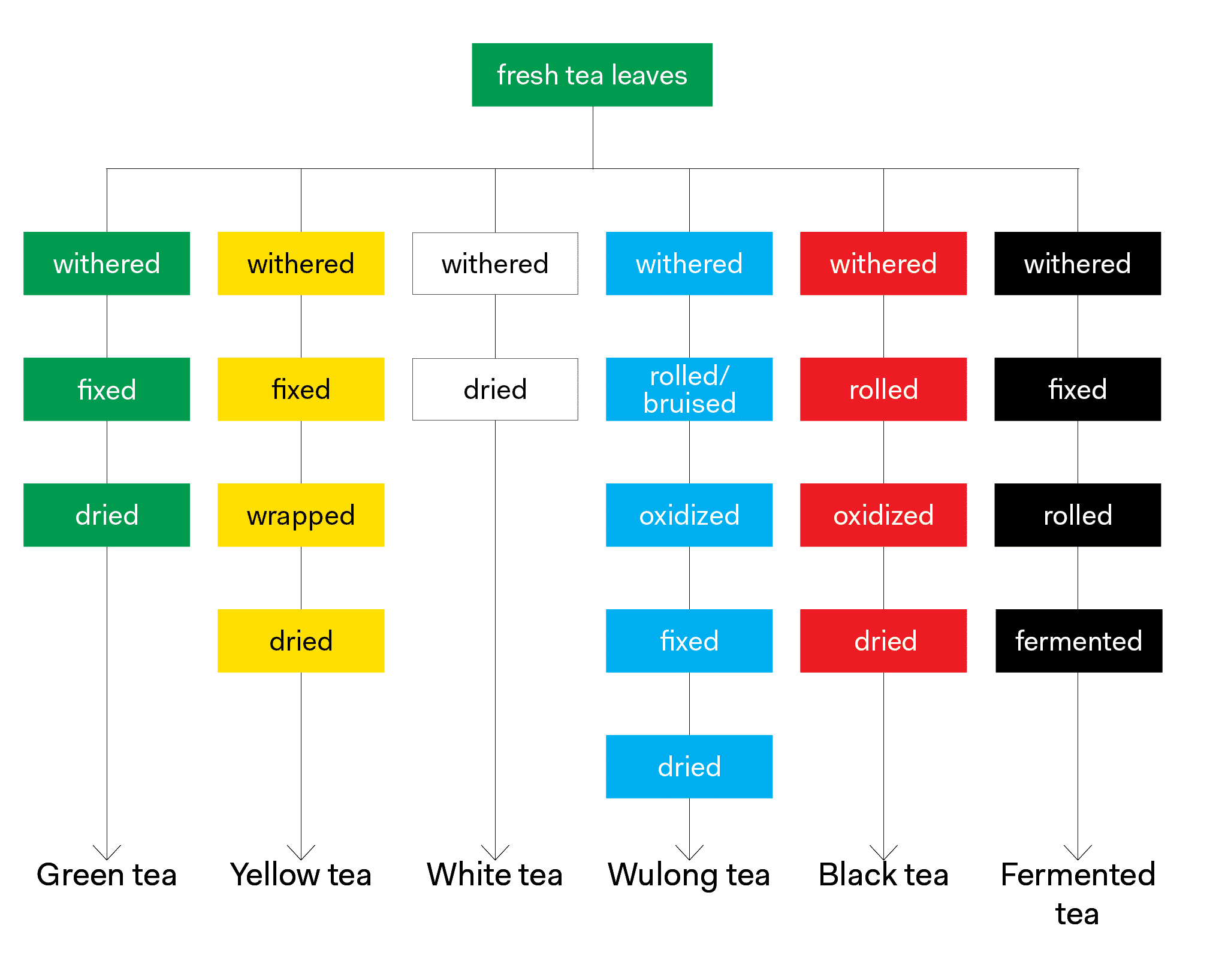
The type of tea being steeped is the most important determinant when it comes to the types and amounts of soluble compounds within tea leaves. Tea types are defined by the processing steps the leaves go through, and thus the resulting chemical components are similar for all styles of finished tea within a type. Across all tea types, the major chemical components in tea leaves fall into the following categories: polyphenols, amino acids, enzymes, pigments, carbohydrates, alkaloids, minerals and volatiles. Individual chemical components lend themselves to a portion of the cup in the form of taste, color or body. The chemical composition of a cup of tea depends not only on the chemical compounds found within the leaves, but also the chemical properties of the water, the surface area of the leaves, the ratio of leaves to water, steeping temperature, and the length of time the leaves are in contact with water.
Water Quality for Tea
Steeped tea is 98% water, which is why using a high quality water source for tea preparation is of utmost importance. But what does it mean for a water to be high quality, or good for tea? There are so many aphorisms that attempt to define how water should be prepared for tea—most of them, pardon my pun, hold no water. Common ones are: use freshly drawn cold water, don’t re-boil water, don’t overboil water, and use water with a high amount of dissolved oxygen. In my experience, these things will do little if anything at all to enhance your tea experience.
For a water source to be suitable for tea preparation we must be sure it tastes good on its own. But what does this mean? Let’s examine the common properties of water, including mineral content, hardness, and pH level, and then discuss ways we can obtain water of ideal quality.
Mineral Content
Good tasting water has a balance of minerals and a clean, even taste. Too high of a mineral content will make water taste tinny and metallic—but a low mineral content will make water taste dull. Mineral content is often erroneously associated with total dissolved solids (TDS), a popular measure for the amount of solid materials dissolved into a water source. The Tea Association of the United States recommends using water with 50 – 150 ppm total dissolved solids to brew the best tea. However, this is not a reliable means for measuring mineral content; TDS is a measure of the total amount of solids that are dissolved in water, regardless of what those solids may be.
Some filtration systems will remove all minerals from water, after which specific minerals are added back in, usually some combination of Magnesium, Calcium, Potassium and Sodium. Each of these minerals are responsible for a portion of the taste in the cup. Unfortunately, the chemistry of how these minerals affect the solubility and dissolution of tea compounds is very complex and not yet fully understood.
Hardness
Water hardness refers to the amount of Calcium and Magnesium dissolved into a water supply. There are two types of hardness: temporary hardness and permanent hardness.
Temporary hardness refers to the presence of Calcium and Magnesium bicarbonates in the water. When in excess, these minerals are known to dull the color of a tea infusion and promote the creation of tea scum. Tea scum is a nasty looking iridescent surface film that will cling to the side of your glass as you drink. Tea scum is caused by the complexation of tea compounds with Calcium Bicarbonate. Temporary hardness can be removed by boiling, but in doing so, it is converted to permanent hardness.
Permanent hardness refers to Calcium and Magnesium carbonates in water; typically water softeners will dissolve these minerals with sodium. Permanent hardness can lead to the deposit of mineral scale in your kettle or water boiler and can result in a stronger, darker brew. It is worth noting that the Langelier Index is a tool that can be used in conjunction with proper filtration to produce a water source that will not deposit scale or cause damage to water heating equipment, but the mechanics of the Langelier Index are beyond the scope of this article.
pH Level
The pH level of water is not a huge concern when finding a water source for your tea. pH levels in tap water typically fall within the EPA’s recommended pH range of 6.5 – 8.5, and any level within that range is fine for tea. A neutral pH is 7. Water with a pH higher than 7 is considered basic or alkaline; use of alkaline water will result in a darker tea infusion and a quicker degradation of tea catechins.1 Water with a pH lower than 7 is considered acidic and will result in a lighter tea infusion.
Choosing a Water Source for Tea
Regardless of your source, the easiest way to be sure that your water is okay for tea preparation is to smell it and taste it. If it is free from odors and unusual tastes, then you may be good to go. Those earnestly searching for a perfect cup of tea will try multiple water sources; you’ll never know how good your tea can be until you try it!
Tap Water
Depending on where you live, tap water can be a great source of water for tea or a terrible source of water for tea. If you are using tap water from a municipal source that has been treated with chlorine, it will affect the taste of the tea. Water that smells and tastes like a swimming pool will produce tea that smells and tastes like a swimming pool! You can remove most of the chlorine in water by boiling it, allowing it to sit in an open container for 24 hours, and/or by filtering it with activated charcoal. Many municipalities have begun replacing chlorine with chloramine, which is not as easy to remove as chlorine. To remove chloramine, you will need to use a special catalytic carbon filter.
If your household’s water source is a well, hardness levels are usually the main issue. I recommend filtering all tap water with an activated carbon filter; pitcher-style filters are widely available, and most of them on the market today will remove any funky smells and tastes from water. For most water types, a simple carbon filter will do the job. The goal is not to filter out all of the naturally-occurring minerals in your water; many minerals in tap water are useful for a proper infusion and desired taste.
Bottled Spring Water
Bottled spring water is extremely environmentally unfriendly, so I recommend using bottled water only as a last resort. Luckily, there isn’t a noticeably huge difference between good filtered tap water and bottled spring water when steeping tea.
When purchasing bottled water, be sure that it is indeed spring water as many bottled waters on the market are merely bottled municipal tap water. Why spring water? Natural springs have a balance of minerals in them, making them ideal for tea preparation. All spring waters contain different levels of different minerals, so it is best to experiment to find one that properly compliments your teas. Just be sure to steer clear of distilled water; this type of water is completely free of minerals and thus tastes dull and flat. Mineral water should also be avoided, as it will often have too high of a mineral content for tea.
Steeping Variables
Surface Area of Tea Leaves
The surface area of tea leaves refers to the total area of the exterior surface of the leaves. For example, let’s take five grams of two finished teas: Sample A is a broken-leaf black tea, and Sample B is a full-leaf, unbroken black tea. Even though we have five grams of both Sample A and Sample B, the surface area of Sample A is greater than that of Sample B because it has more exposed leaf parts due to its broken nature. When steeped, Sample A will have a higher area in contact with water, allowing for more soluble compounds to dissolve into the water at the same time. This means that when steeping a tea that has a high surface area (such as Sample A), you will achieve a beverage of desired strength faster than you would with a tea with a lower surface area (Sample B).
Ratio of Tea Leaves to Water
The amount of tea leaves used during steeping affects the strength of the tea liquor. Adding more leaves to the same amount of water increases the overall surface area of the tea, literally making more tea compounds available for dissolution. As many tea drinkers know, the fastest way to achieve a stronger brew is to steep more tea leaves. However, you can still reach a beverage of desired strength with a smaller amount of leaves; it will simply require a longer steeping time.
Steep Time
Steep time refers to the amount of time the leaves are in contact with water—the amount of time that dissolution can occur. If given enough time, some compounds in the tea leaves would reach equilibrium, meaning that the concentration of these compounds in the leaves would be the same as the concentration of the same compounds in the water. This wouldn’t necessarily make for a palatable infusion, so we adjust the steep time to control the concentration of the compounds in our cup.
Water Temperature
Temperature is related to kinetic energy. Increasing the water temperature increases the kinetic energy among the water molecules, allowing them to more effectively dissolve solute molecules. Thus, an increase in water temperature increases the rate of dissolution of soluble compounds in tea leaves. The solubility of various teas peaks at different temperatures; the chemical composition of a cup of tea steeped at different temperatures will vary, sometimes greatly, and so will its taste. In the case of solids, solubility increases and the rate of dissolution increases as the water temperature increases.
Steeping Vessel
The kinetics of steeping work irrespective of the vessel that the steeping occurs in. However, when choosing a vessel, there are two things that you must take into consideration:
- How much heat will the vessel absorb away from your water?
- Do you have a way to quickly separate the leaves from the water to halt the steeping process?
After you have poured your hot water into a steeping vessel, the temperature can quickly decrease. Depending on the mass of your vessel and the thermoconductivity of its material, the temperature of tea can decrease up to fifteen degrees after thirty seconds. However, this effect can be greatly reduced by preheating the vessel. To preheat your vessel, simply fill it with water at or slightly above the desired steep temperature. Next, let it sit for thirty seconds and decant the water. Preheating reduces the decrease of temperature after thirty seconds by half.
Second, it is important that you are able to quickly separate the leaves from the water to halt the steeping process. The simplest way to halt the steeping process is to decant the vessel’s contents through a strainer. Some steeping vessels are equipped with a built-in strainer that can be removed once steeping is complete.
Relationship Between Time and Temperature
When steeping tea, the results of water temperature and steep time on our beverage are inversely correlated. This means that if you have a preferred steep time and water temperature that you use to prepare an infusion of a particular strength, slightly increasing the steep time and decreasing the water temperature will yield a similar result. Likewise, decreasing the steep time and increasing the water temperature will also yield a similar result.
I have observed that for each 20 degree rise in steep temperature, you can halve the infusion time. Likewise, for each 20 degree decrease in steep temperature you can double the infusion time and achieve a similar result. Remember though that changing the temperature will alter the solubility of the chemical compounds in the leaves. Higher temperatures can result in cups of tea with differing chemical compositions; while adjusting time can increase or decrease the overall strength of the beverage, adjusting temperature will slightly affect taste.