The bulk of tea produced in the world is commodity tea, meaning that it is actively traded and it’s price is determined by the markets. Commodity tea is relatively cheap, with the worldwide average price of commodity black tea typically in the area of $2.85USD/kilogram.
Many of the world’s famous tea cultures1 are famous because they are promulgated by common man and are thus largely based on cheap commodity tea. There is however, a larger amount of high quality tea being produced every year, what some are calling the specialty tea industry.
One way to think about the difference between commodity tea and specialty tea is that commodity tea production prioritizes quantity over quality and specialty tea production prioritizes quality over quantity.
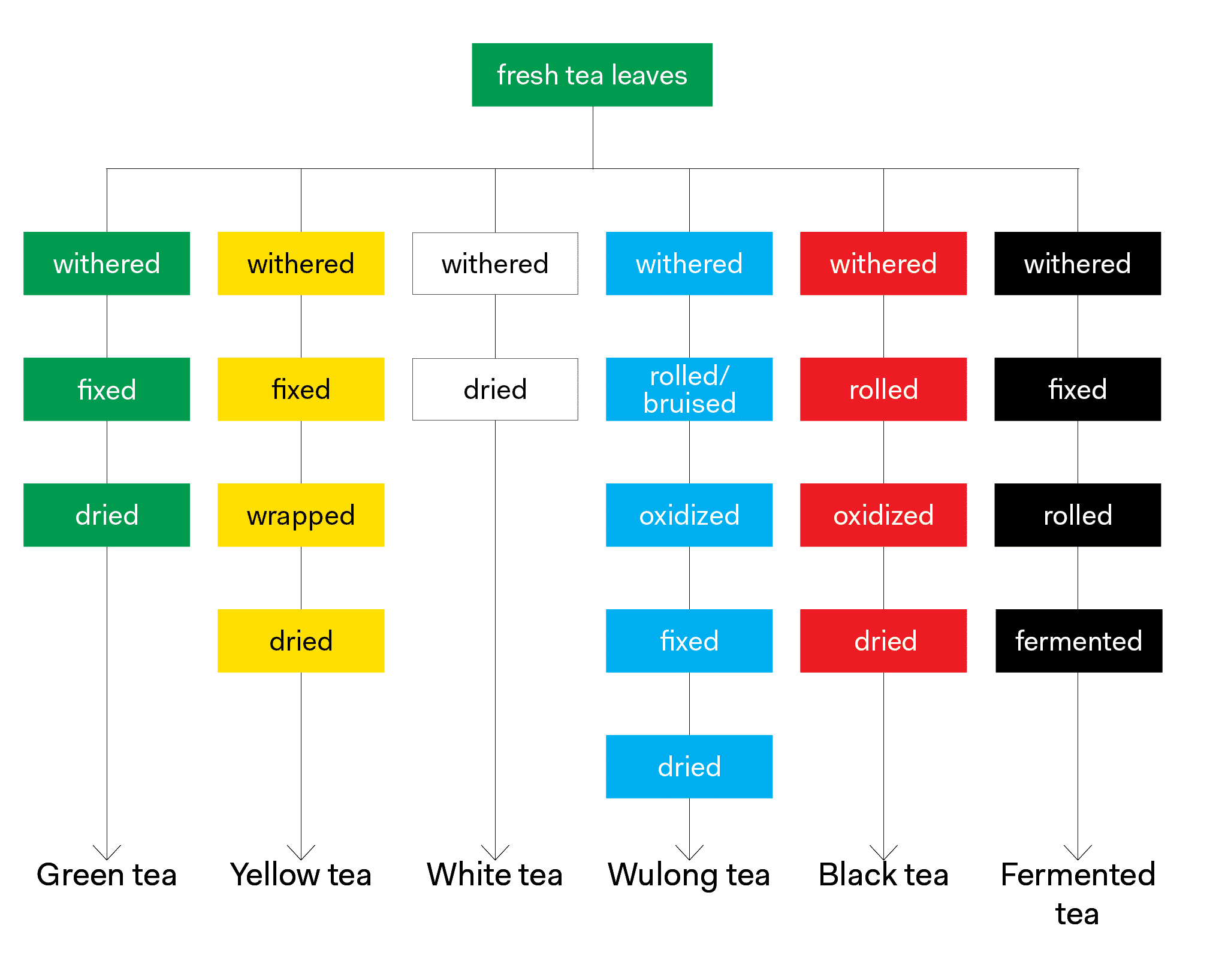
What is quality? Leaf format is arguably the most important determining factor of quality for a tea and is inherently a function of the processing methods used to create it. In general2, lower quality (commodity) teas tend to be finely ground or made up of leaf particles very small in size, while higher quality (specialty) teas tend to be made up of full leaves or perhaps even more than one leaf and/or bud.
For this article, I’ll assume that you have acquired a high quality full-leaf tea. Let’s look at the factors that make for an exemplary cup of it.
Tea (the beverage) is made by infusing tea leaves in a solvent (water) to make a solution wherein the solute is made up of the soluble solids within the tea leaves. The resulting solution is on average 98% water and 2% compounds from within the leaf. The tea equation is made up of the soluble solids3 within the leaves (which can be generalized by type of tea), the surface area of the tea, the chemical properties of the water, atmospheric pressure, water temperature, and infusion time.
Type of Tea
The type of tea being steeped is the most important determinant when it comes to the types and amounts of soluble solids within tea leaves. Tea types are defined by the processing steps the leaves go through and thus, the resulting chemical components in the leaves are similar for all styles of tea within a type.
Across all tea types, the major chemical components in tea leaves fall into the following categories: polyphenols, amino acids, enzymes, pigments, carbohydrates, alkaloids, minerals and volatiles. Individual components in each category lend themselves to a portion of the cup in the form of taste, color or body. The rate of dissolution of those components that are soluble in water depends on the surface area of the tea leaves, the chemical properties of the water, atmospheric pressure, water temperature, and infusion time.
Surface Area of Tea Leaves
The rate of dissolution of the soluble components in tea leaves increases with the surface area of the leaves. Thus, a ground tea with fine leaf particles will impart flavor and color in water quicker than an unbroken, full-leaf tea. Adding more full-leaf tea increases overall surface area within the solvent and will also increase the rate of dissolution.

One gram each of CTC Assam black tea (left), Sun Moon Lake black tea (middle), and Ball-style Taiwan Oolong (right). Despite the fact that the same amount of each tea is pictured, the surface area of each varies greatly.
Chemical Properties of Water
The leaching qualities of water on a molecular level are either aided or impeded by pH levels, hardness levels, and mineral levels (often measured by TDS or total dissolved solids).
The pH level of drinking water should not be a huge concern when finding a water source for your tea, this is because pH levels will typically fall within the EPA’s recommended pH range of 6.5 – 8.5, any level within being fine for tea. A neutral pH is 7, water with a pH higher than 7 is considered basic or alkaline and will result in a darker tea infusion. Water with a pH lower than 7 is considered acidic and will result in a lighter tea infusion.
Water hardness refers to the amount of calcium and magnesium dissolved into a water supply. There are two kinds of hardness: temporary hardness and permanent hardness. Temporary hardness refers to the presence of calcium and magnesium bicarbonates and is known to dull the color of a tea infusion and promote the creation of tea scum. Tea scum is a nasty looking iridescent surface film on tea that will cling to the side of your glass as you drink it. Temporary hardness can be boiled out of the water – in doing so, it is converted to permanent hardness. Permanent hardness refers to calcium and magnesium carbonates in water, typically water softeners will dissolve these minerals with sodium. Tea polyphenols bind to the minerals in hard water resulting in a stronger and darker brew.
Good tasting water has a balance of minerals and a clean, even taste. Too high of a mineral content will make water taste tinny and metallic, too low and water tastes dull. Mineral content is often erroneously associated with total dissolved solids or TDS, a popular measure for the amount of solid materials dissolved into a water source. The Tea Association of the United States recommends using water with 50 – 150 ppm total dissolved solids to brew the best tea. However, this is not a reliable means for measuring mineral content as TDS is a measure of the total amount of solids that are dissolved in water, regardless of what those solids may be.
While TDS alone is not a reliable marker as to the quality of a water source, it is known that some levels of minerals in water are beneficial and actually enhance the flavor of tea. Too high of a mineral content and a tea will taste metallic, conversely, distilled water, void of minerals produces a flat tasting tea. Many tea and coffee shops will filter everything out of their water supply then add calcium, potassium, and sodium back into the water.
Atmospheric Pressure
Atmospheric pressure at sea level is around 14.7 psi, the boiling point there is 100°C. Below sea level, where atmospheric pressure is higher, the boiling point of water rises above 100°C. Above sea level, where the atmospheric pressure is lower, the boiling point of water dips below 100°C. This effect is exacerbated in several newfangled devices such as the bkon tea brewer which infuses tea in a vacuum. This low pressure environment allows boiling to occur at a temperature well below 100°C. Visit Engineering Toolbox to see a chart of boiling points at different levels of pressure.
Water Temperature
In general, heat increases the rate of dissolution of any solute in a solvent. A pattern you may have noticed is that most tea merchants recommend using cooler water for teas that are less oxidized (green teas, yellow teas, white teas, oolong teas) and hotter water for teas that are more oxidized (black teas, post-fermented teas). The main reason for this pattern is polyphenol content. During oxidation, catechins (a group of polyphenols) are converted to theaflavins and thearubigins (two other groups of polyphenols). These polyphenols, especially the catechins, are known for their bitterness and astringency and hot water draws them out of the leaves into solution more easily. Theaflavins and thearubigins present in oxidized teas not only contribute to taste but also are a large part of the color of the liquor.
To recap, a general rule of thumb (keeping the infusion time the same) is that teas that have a lower level of oxidation inherently have a higher level of catechins and taste better when steeped with cooler water. Teas that have a higher level of oxidation inherently have a lower level of catechins and taste better when steeped with hotter water. Note that this is completely overly simplified, and that polyphenols, while they do make up a large proportion of soluble tea solids, are only one of the hundreds of compounds found in tea. Here’s a neat and nerdy trick to use to obtain water that is a specific temperature.
Ratio of Tea Leaves to Water
As a starting point, I recommend using 1 gram for each 50ml of water for tea preparation. Remember, because styles of tea within a type are processed in a similar manner, their chemical composition is also relatively similar. If you steep similar styles of tea in the same way, it should produce a similar cup.
Infusion Time
Infusion time and temperature go hand in hand. You can obtain a similar color and taste of infusion steeping a tea at a hotter temperature for a shorter amount of time than you can steeping the same tea at a lower temperature for a longer amount of time. As different chemical components in the leaf are more or less soluble at different temperatures, results may vary, but I’ve done several dozen rounds of trials and found similar results with each of these infusion times and temperatures for green, white, oolong and black teas using the reccomended dosage above. The optimal infusion time and temperature for each according to my taste preference has been highlighted in green4:
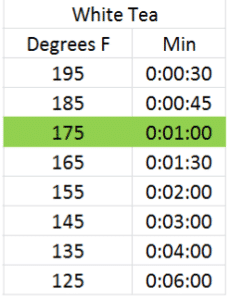
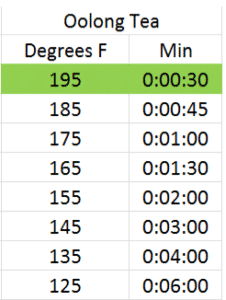
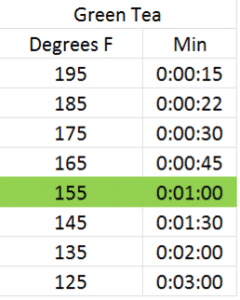
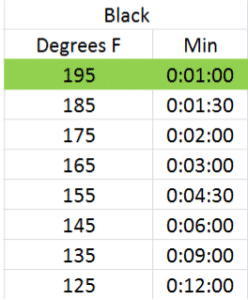
Basically, for each 20 degree rise in steeping temperature, the infusion time halves. Give it a try and see what you think.
Notes:
- A list of world famous tea cultures would likely include: British Afternoon Tea, Indian Masala Chai, Moroccan Mint, Kashmiri Kahwah and many others.
- This is a generalization as matcha is ground and can be high quality.
- A large part of tea flavor comes from the volatile aroma compounds which exist in trace amounts. Volatile here means that these compounds are no longer solid.
- The optimal times and temperatures were derived using a blind taste test of 10 different full-leaf pure teas from each category.